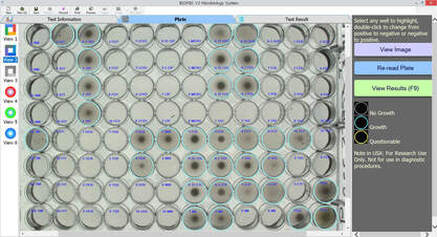
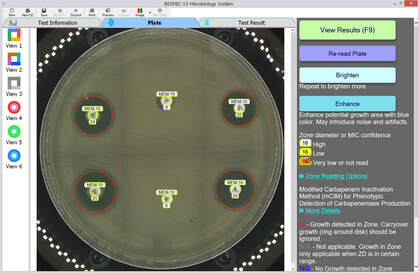
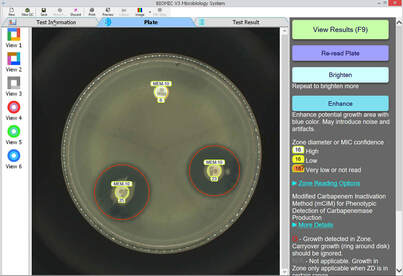
Current AMR surveillance programs sponsored by the US-FDA and CDC use BIOMIC V3 systems to digitally read, interpret, and record standard AMR tests.
View the BIOMIC V3 website here AMR Testing Modules with BIOMIC V3:
AMR Advantages with BIOMIC V3:
- Open system with no proprietary consumables
- No routine system maintenance - Portable: Weight: 16kg, Size: 51 x 53 x 38cm - Operational in extreme environments - Disk ZD & MIC, Strip MIC, 96-Well MIC - Annual CLSI & EUCAST guidelines & expert rule updates - Standardized test procedures and data - Custom epidemiology & QC reports - Verified QC & QA testing - Alerts for unusual, unlikely, or rare test results - Training videos for all procedures - Custom data export formats - Test plate images saved - PC Windows based software - Installation & calibration in less than 1 hour - Software in 12 languages |
AMR Current Programs with BIOMIC V3:
CDC: Carbapenem Resistance (CRE)** Learn More >
CDC: Gonococcal Isolate Surveillance Program Learn More >
US-FDA: Vet-LIRN Veterinary Laboratory Investigation and Response Network Learn More >
US-FDA: NARMS National Antimicrobial Resistance Monitoring System Learn More >
USDA: NAHLN National Animal Health Laboratory Network Learn More >
AMR Previous Program with BIOMIC V3:
From 1997-2010, 151 hospital labs in 40 countries with BIOMIC V3 systems participated in the ARTEMIS drug resistance study sponsored by Pfizer Pharmaceuticals. 50+ journal publications demonstrate BIOMIC V3's testing and AMR surveillance capabilities. View publications >
AMR Multi-Lab Networks with BIOMIC V3:
Multiple BIOMIC V3 systems can be connected on a global network for hospital or private lab groups, government MOH programs, or multiple lab studies. Sites may collect test and QC results to monitor AMR trends from a central location. Study results may be used for regulatory filing, research, or marketing purposes.
CDC: Carbapenem Resistance (CRE)** Learn More >
CDC: Gonococcal Isolate Surveillance Program Learn More >
US-FDA: Vet-LIRN Veterinary Laboratory Investigation and Response Network Learn More >
US-FDA: NARMS National Antimicrobial Resistance Monitoring System Learn More >
USDA: NAHLN National Animal Health Laboratory Network Learn More >
AMR Previous Program with BIOMIC V3:
From 1997-2010, 151 hospital labs in 40 countries with BIOMIC V3 systems participated in the ARTEMIS drug resistance study sponsored by Pfizer Pharmaceuticals. 50+ journal publications demonstrate BIOMIC V3's testing and AMR surveillance capabilities. View publications >
AMR Multi-Lab Networks with BIOMIC V3:
Multiple BIOMIC V3 systems can be connected on a global network for hospital or private lab groups, government MOH programs, or multiple lab studies. Sites may collect test and QC results to monitor AMR trends from a central location. Study results may be used for regulatory filing, research, or marketing purposes.
About Giles Scientific:
Since 1984, Giles
Scientific has served clinical and industrial microbiology labs in 100
countries. The BIOMIC V3 Microbiology System automates the reading and interpretation of microbiology tests utilizing
color digital imaging technology. Giles Scientific is an ISO 9001:2015 and ISO
13485:2016 certified company. Our systems are designed and manufactured in
Santa Barbara, California, USA.